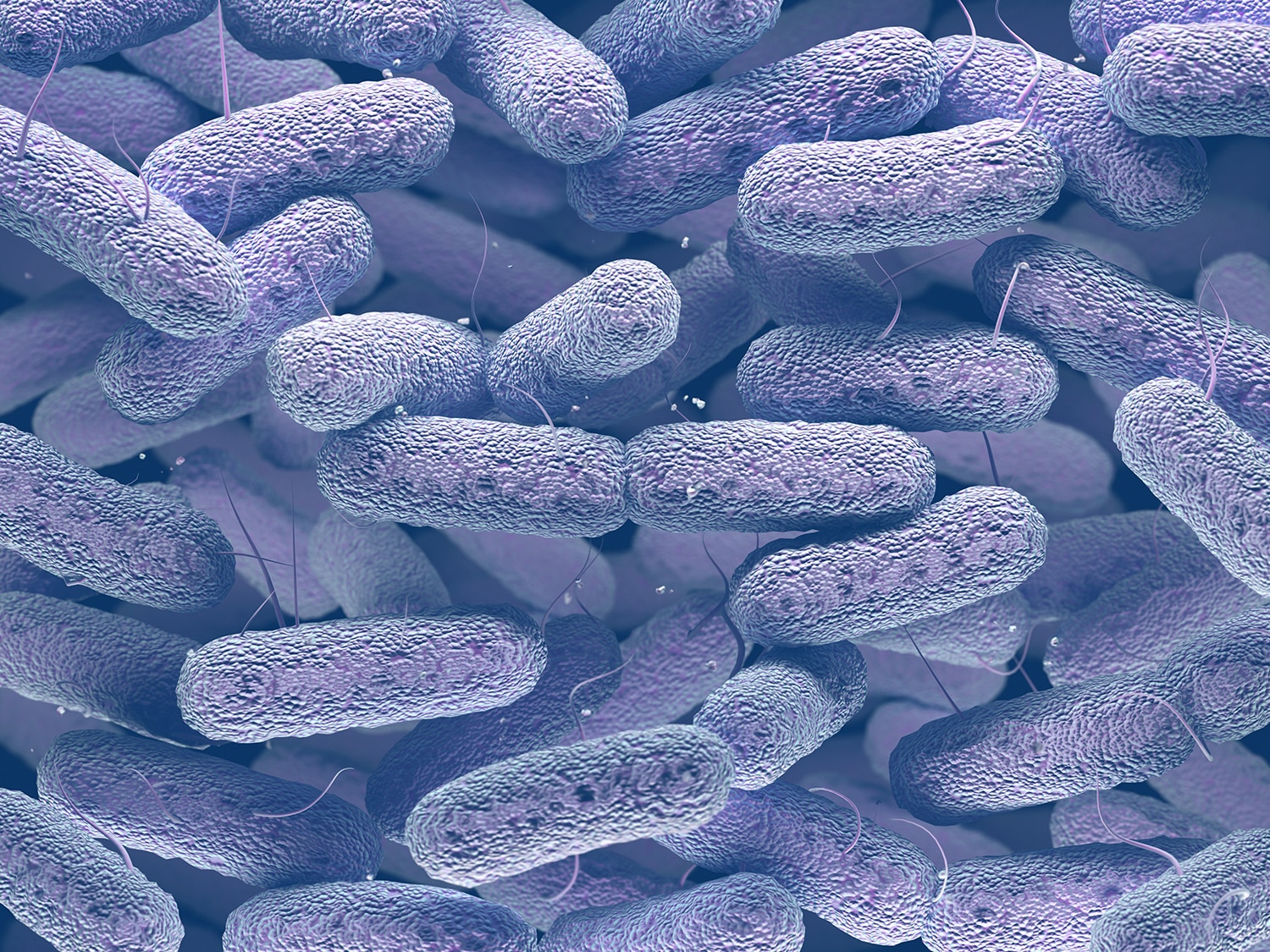
How Gut Microbes Could Increase Cancer Risk
Researchers have found that the microbes in a person’s gut influence colorectal cancer risk, but it’s been difficult to pin down how this is happening. A paper published Feb. 27 in Nature explains one possible mechanism for how bacteria could increase cancer risk. Researchers exposed gut tissue in the lab to a strain of Escherichia coli that produced a substance called colibactin. The researchers found that the gut tissue exposed to the bacteria developed a distinctive signature of mutations. They found the same pattern of mutations in the tumors of some patients with colorectal cancer, indicating that bacteria that secrete colibactin could be one cause of increased cancer risk. “More than 5% of colorectal cancer had high levels of the footprint, while we only saw it in less than 0.1% of all other cancers,” study co-author Jens Puschhof said in a press release. “Imagine studying a gut bacterium’s footprint for months in a dish, and then finding back the same footprint in the DNA of patients.”
Appendix Cancer on the Rise
Cancer of the appendix is rare, but a study published Feb. 26 in Cancer shows that the rate of this cancer is increasing in the U.S. and Canada. The researchers found that between 2000 and 2016, the rate of appendix cancer rose 232% in the U.S. and 292% in Canada. Previous research showed that appendix cancer increased in the U.S. between 2000 and 2009, but questions remained about whether doctors were simply finding more appendix cancers during surgeries to remove the appendix. The authors of the new study looked at the rate of appendectomy in Canada over time, finding that it was stable. This indicates that that some other, unknown factor is leading to the rise in appendix cancer cases.
Expanded Approval for a Breast Cancer Drug
The Food and Drug Administration on Feb. 25 expanded its approval of Nerlynx (neratinib) to apply to some patients with advanced or metastatic HER2-positive breast cancer. The drug is approved for use in combination with the chemotherapy drug capecitabine in patients who have previously been treated with two other therapies targeting HER2. Nerlynx previously was approved for treatment of some patients with early-stage HER2-positive breast cancer.
Cancer Today magazine is free to cancer patients, survivors and caregivers who live in the U.S. Subscribe here to receive four issues per year.