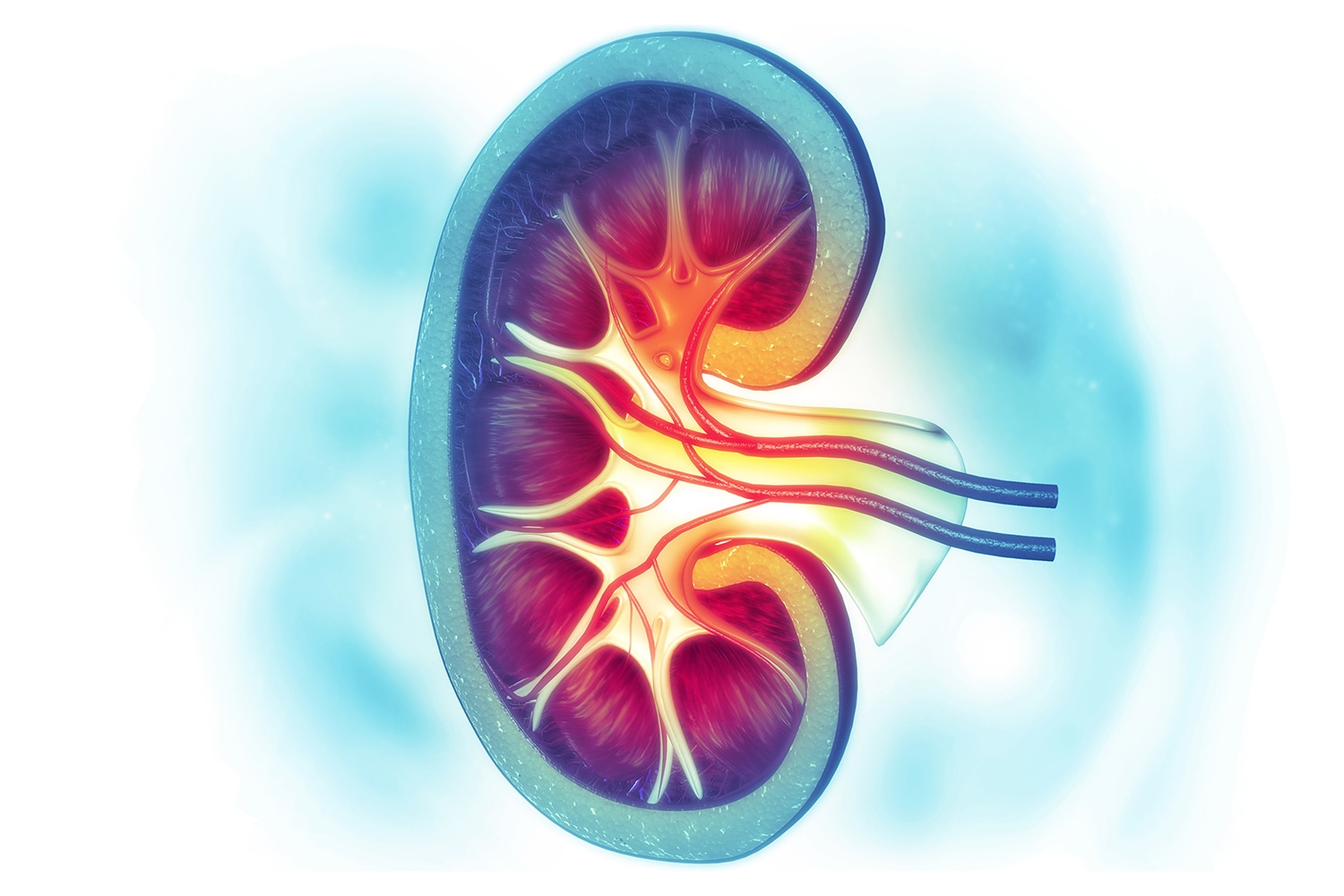
Combining Targeted Therapy and Immunotherapy
The New England Journal of Medicine on Feb. 16 published two papers on combinations of immunotherapy and targeted therapy for treatment of advanced kidney cancer, finding that these combinations are superior to using a targeted therapy alone. One trial compared Sutent (sunitinib), a targeted therapy that inhibits the growth of blood vessels, to a combination of the immunotherapy drug Keytruda (pembrolizumab) and Inlyta (axitinib), which also inhibits blood vessel growth. The second trial compared Sutent to Inlyta plus another immunotherapy drug, Bavencio (avelumab). “Both combinations are expected to become new standards of care and to be incorporated into future guidelines,” Bernard Escudier of the Gustave Roussy Cancer Campus in Villejuif, France, wrote in an editorial published in the same journal. Escudier points out that a combination of two immunotherapy drugs, Opdivo (nivolumab) and Yervoy (ipilimumab), has also been shown to be superior to just Sutent in patients with advanced kidney cancer.
A Genetic Smoking Gun
The U.S. Food and Drug Administration said last fall that it will seek to ban menthol cigarettes. A study published Feb. 15 in PLOS Genetics finds that use of these mint-flavored cigarettes is more common in people who have a specific genetic variant that is only present in people with African ancestry. The variant may make menthol cigarettes feel less irritating to the airway, the researchers say, and was present in the DNA of 5 to 8 percent of the African-Americans in the study. More than 80 percent of African-Americans who smoke use menthol cigarettes, while only 30 percent of white American smokers use this type of cigarette. Advertising for menthol cigarettes has historically targeted African-Americans. “While this gene variant can’t explain all of the increased use of menthol cigarettes by African-Americans, our findings indicate that this variant is a potentially important factor that underlies the preference for menthol cigarettes in this population,” said study co-author Dennis Drayna of the National Institute on Deafness and Other Communication Disorders in a press release.
What Prevents Patients From Joining Trials?
A review study published Feb. 19 in the Journal of the National Cancer Institute looks at the barriers that keep cancer patients from joining clinical trials. Across 13 U.S. studies of cancer clinical trial enrollment, nearly 56 percent of patients were not being treated at a center with a trial available for their cancer type and stage. An additional 22 percent were ineligible to join available trials for their cancer type and stage. About 8 percent of patients did enroll in a trial. That leaves just under 15 percent of patients who were eligible for a trial but didn’t join one, whether because their doctor didn’t bring it up or because they decided not to participate. “The reason most patients don’t go into clinical trials is not because of the patient, but because of all the barriers in front of the patient, including structural and clinical factors,” study co-author Joseph Unger of the Fred Hutchinson Cancer Research Center in Seattle told Reuters.
Treatment Shortage
There’s a shortage of a drug used to treat bladder cancer, reports STAT. Bacillus Calmette-Guérin, or BCG, is an immunotherapy that has been used to treat bladder cancer since the 1970s, the article explains. “This BCG shortage is a huge deal,” Robert Abouassaly, a urologist at the Cleveland Clinic, told STAT. “We don’t have any alternatives that are as effective for these patients.” Merck, which manufactures the drug for the U.S. and Europe, says it is making as much of the drug as it currently can.
Cancer Today magazine is free to cancer patients, survivors and caregivers who live in the U.S. Subscribe here to receive four issues per year.